
Pharmaceutical architecture, flows and HVAC systems
Where critical deviations are born (or avoided) In the previous article in this series of three technical publications we addressed

Where critical deviations are born (or avoided) In the previous article in this series of three technical publications we addressed

In the pharmaceutical industry, the design of a facility is neither an architectural exercise nor mere regulatory compliance. Is

In the development of new facilities, manufacturing lines, automated systems or critical equipment in regulated environments, delays and

Aseptic manufacturing represents one of the most critical environments within the pharmaceutical industry. The absence of a

The pharmaceutical industry faces a historic trilemma: reduce time to market, maintain quality levels

In the pharmaceutical industry, robust control of equipment, facilities and processes is essential to guarantee product safety.
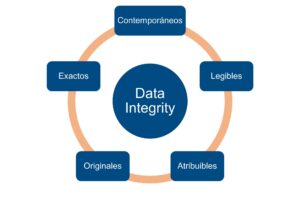
Data Integrity is a central pillar in the pharmaceutical industry and in any regulated environment.

The design of clean areas for aseptic processes is one of the pillars on which the manufacturing of

Regulatory compliance and contamination control strategies 1. Introduction to the modern pharmaceutical engineering paradigm The pharmaceutical industry